Bacteriophages found to combat antibiotic resistance in kitchen sponges
Researchers at the New York Institute of Technology have discovered that bacteriophages that combat antibiotic resistance may be present in household kitchen sponges. As the threat of antibiotic resistance grows, bacteriophages, or phages for short, may prove useful in fighting bacteria that cannot be killed by antibiotics alone. The research will be presented at ASM Microbe, the annual meeting of the American Society for Microbiology.
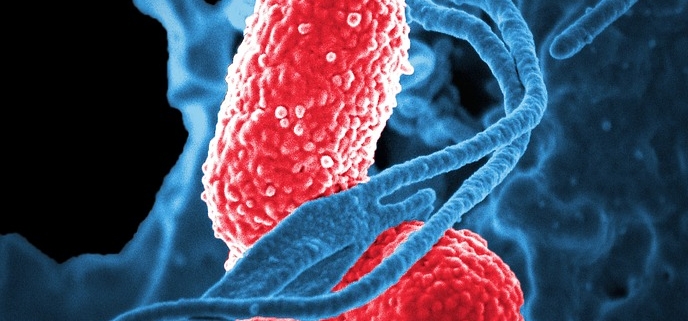
Antibiotic resistance is on the rise, and bacteriophages are considered a potential therapy against multi-resistant bacterial strains.
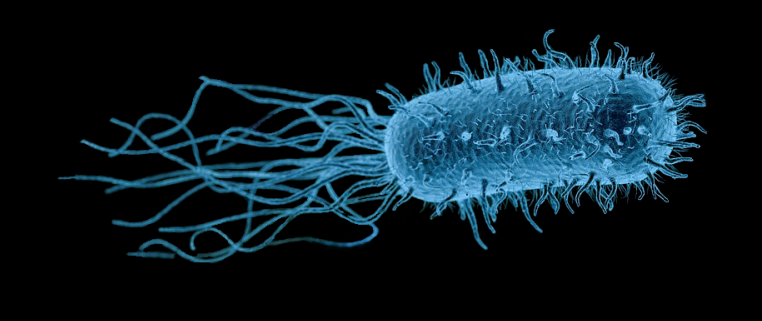
Antibiotic resistance is one of the greatest threats to global health, food security, and development today. Bacteriophages—or phages—are viruses that target and kill bacteria. They live wherever bacteria are found, including soil and water, and are being studied as an alternative treatment for drug-resistant infections.
A kitchen sponge is exposed to all types of microbes, forming a vast microbiome of bacteria. Phages are the most abundant biological particles on the planet and are typically found wherever bacteria reside. With this understanding, kitchen sponges seemed a likely place to find them.
This project began as part of a research study with seven students at the New York Institute of Technology (NYIT) in Old Westbury, New York.
Students in a research class isolated bacteria from their own used kitchen sponges and then used the bacteria as bait to find phages that could attack them. Two students successfully discovered phages that infect bacteria living in their kitchen sponges. “Our study shows the value of searching in a microbial environment where potentially useful phages may exist,” said Brianna Weiss, a life sciences student at the New York Institute of Technology.
Source and more at: https://speciality.medicaldialogues.in/bacteriophages-key-to-counter-antibiotic-resistance-found-in-kitchen-sponge/amp/