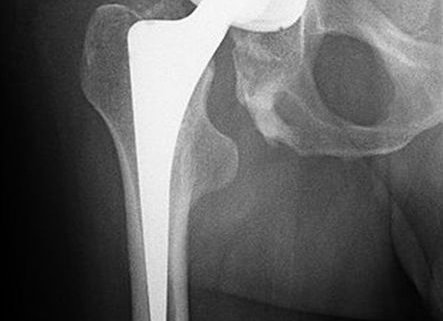
Bacteriophage therapy for a chronic MRSA prosthetic joint infection
This is the case of a 72-year-old man with a chronic methicillin-resistant Staphylococcus aureus prosthetic joint infection. After the third intravenous bacteriophage therapy dose, an unusual, reversible transaminitis led to the discontinuation of the bacteriophage therapy. Nevertheless, the treatment was successful, and the patient’s severe chronic infection was eradicated.
According to the findings of this research team, this is the first case of bacteriophage therapy successfully used as an adjuvant therapy to cure a chronic MRSA PJI. Two other successful bacteriophage treatments of MSSA and Pseudomonas PJI have been reported [10,11]. Unique to our case is that no chronic suppressive antibiotics were used. Little is known about the optimal treatment duration or administration routes for PJI. It was planned to treat for longer, but the therapy was stopped when significant transaminitis occurred. Nevertheless, successful sterilization of the patient’s joint and devitalized bone was achieved with one IA and a 3-day IV bacteriophage therapy in combination with standard-of-care IV antibiotics over 6 weeks. Given the ability of bacteriophages to self-replicate, perhaps only a few days of bacteriophage therapy are required as a supplement to surgical debridement. Clinical trials are needed to determine an appropriate duration of bacteriophage therapy under these conditions [10,11].
To date, all PJI patients successfully treated with bacteriophages have required surgical debridement [10,11]. This surgery allows for manual scrubbing of the biofilm, ensures the prosthesis is salvageable, and enables the instillation of bacteriophages directly onto the biofilm. Local dosing of bacteriophages may be vital for the elimination of biofilm infections, but limited data exists beyond case reports [10,11,12]. No adverse events occurred with repeated IA doses, possibly due to limited systemic absorption. Future studies must be conducted to determine appropriate administration routes in PJI.
The most unique aspect of our case was the transaminitis that occurred after the third IV bacteriophage dose. This appeared to have been caused by the bacteriophage therapy. No other liver function disorder was identified, and the transaminitis was reversible and not life-threatening. Figure 2 shows liver function during the course of the bacteriophage therapy.
The patient had hepatomegaly, but non-alcoholic fatty liver disease could not be radiographically proven, and the biopsy was postponed. Over 99% of IV bacteriophage therapy is rapidly degraded by the liver and spleen [13,14,15]. This research team’s theory is that the underlying steatosis caused the liver macrophages to trigger a dysregulated local cytokine response when confronted with a large number of bacteriophages that required hepatic clearance. This local reaction may have led to inflammatory changes in the hepatocytes, resulting in an increase in AST and ALT. This is supported by studies examining the role of liver macrophages in steatosis and older studies on the hepatic clearance of bacteriophages [13,14,15]. It is not known whether mild to moderate elevations in liver enzyme levels frequently occur after administration of bacteriophages. Furthermore, it is not known whether continued IV bacteriophage administration would have worsened the transaminitis or led to adaptation and resolution. For now, intravenously administered bacteriophages should be used with caution in patients with underlying liver pathology, and liver enzymes should be closely monitored. This case is limited as we did not examine our patient’s cytokine response to the bacteriophage therapy. Future studies should evaluate this response to learn more about the normal human cytokine response to bacteriophage therapy.
In summary, salvage of the patient’s prosthesis was not possible due to severe bone erosion. However, we were able to sterilize the patient’s severe chronic MRSA PJI with a single virulent bacteriophage administered IA and IV for three days in combination with IV antibiotics. Further PJI studies are required to determine the optimal duration and mode of phage administration. Bacteriophage therapy has enormous potential for curing PJI, but phase 1 and 2 clinical trials must be conducted.
Translated from the source:
https://www.mdpi.com/2079-6382/9/5/241/htm
Salvage Bacteriophage Therapy for a Chronic MRSA Prosthetic Joint Infection
by James B. Doub,*,Vincent Y. Ng, Aaron J. Johnson,Magdalena Slomka, Joseph Fackler, Bri’Anna Horne ,Michael J. Brownstein ,Matthew Henry,